Genetic Myopathies and Therapeutic Development
Group 1, led by E. Malfatti, focuses on the identification and in-depth characterization of new genetic myopathies and on the development of therapeutic strategies for these rare diseases.
A major objective is the establishment of patient registries and natural history studies, which are essential prerequisites for future clinical trials.
In parallel, the group continues the clinical, histopathological, and genetic characterization of a large cohort of patients followed at the Henri Mondor Neuromuscular Reference Center of the FILNEMUS network (Henri Mondor website – CRMRNM), with the aim of identifying new pathogenic genes. In the absence of pharmaceutical interest in rare myopathies, the group is actively developing innovative therapeutic strategies, notably a gene therapy targeting MYPN-related myopathy using an AAV vector, tested in vitro on 3D myofiber models and in vivo in myopalladin knock-out mice. Drug repurposing strategies using patient-derived hiPSC models are also being developed to identify actionable therapeutic targets for rare genetic congenital myopathies (CMYO).
These integrated approaches aim to link genetic discovery, disease modeling, and therapeutic translation for rare genetic myopathies.
Reference:
1Malfatti E, Caramizaru A, Lee H, Kim J, Shoaito H, Pennisi A, Souvannanorath S, Authier FJ, Dumitrescu A, Fahmy N, Escobar-Cedillo RE, Miranda-Duarte A, Luna-Angulo AB, Nouioua S, Benchaabi O, Tazir M, Hallal S, Martinez P, Castiglioni C, Dobrescu A, Tajsharghi H. NEUROMYODredger: Whole Exome Sequencing for the Diagnosis of Neurodevelopmental and Neuromuscular Disorders in Seven Countries. Clin Genet. 2025 Feb 25;108(3):318-22. doi: 10.1111/cge.14736.
2Cardone N, Taglietti V, Baratto S, Kefi K, Periou B, Gitiaux C, Barnerias C, Lafuste P, Pharm FL, Pharm JN, Panicucci C, Desguerre I, Bruno C, Authier FJ, Fiorillo C, Relaix F, Malfatti E. Myopathologic trajectory in Duchenne muscular dystrophy (DMD) reveals lack of regeneration due to senescence in satellite cells. Acta Neuropathol Commun. 2023 Oct 19;11(1):167. doi: 10.1186/s40478-023-01657-z.
3 Onnée M, Malfatti E. The widening genetic and myopathologic spectrum of congenital myopathies (CMYOs): a narrative review. Neuromuscul Disord. 2025 Apr;49:105338. doi: 10.1016/j.nmd.2025.105338.
Acquired Neuromuscular Diseases and Immune-Mediated Myopathies
Group 2, led by F.J. Authier, studies the pathophysiology and treatment of acquired neuromuscular diseases through five main axes: immune-mediated myopathies, interferonopathies, acquired ciliopathies, chronic fatigue syndromes, and COVID-19–related muscle involvement.
Healthy skeletal muscle is continuously exposed to mechanical stress, infectious agents, and xenobiotics, which may trigger inflammation, oxidative stress, or metabolic dysfunction. Inappropriate responses can lead to structural and functional alterations of muscle. The group aims to decipher the molecular mechanisms underlying these disorders in order to improve diagnosis and develop innovative therapeutic approaches.
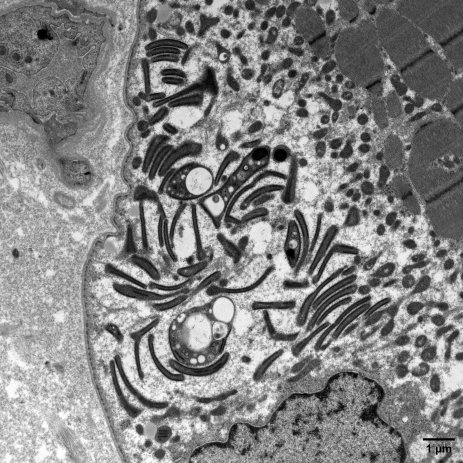
Particular emphasis is placed on interferon-induced diseases, such as juvenile dermatomyositis and inclusion body myositis (IBM). In IBM, persistent infiltration of CD8 T lymphocytes secreting IFNγ leads to progressive muscle degeneration and resistance to immunosuppressive treatments. Ongoing work includes a clinical trial evaluating the JAK inhibitor ruxolitinib and studies exploring the role of the primary cilium. Advanced transcriptomic approaches and spatial omics are used to revisit the pathophysiology of inflammatory and dysimmune myopathies.
Through clinical trials, large-scale data analyses, and mechanistic studies, this research aims to identify new therapeutic targets and improve the prognosis of patients with acquired neuromuscular diseases, notably through the BIGTIM clinical trial, a Phase IIb trial evaluating interferon-γ blockade with ruxolitinib in inclusion body myositis, whose primary objective is to improve overall locomotor performance after one year of treatment, measured by an increase greater than 60 m in the 6-minute walk test (6MWT).
Reference:
1Martin L, Kaci N, Benoist-Lasselin C, Mondoloni M, Decaudaveine S, Estibals V, Cornille M, Loisay L, Flipo J, Demuynck B, de la Luz Cádiz-Gurrea M, Barbault F, Fernández-Arroyo S, Schibler L, Segura-Carretero A, Dambroise E, Legeai-Mallet L.
Theobroma cacao improves bone growth by modulating defective ciliogenesis in a mouse model of achondroplasia. Bone Research. 2022;10(1):8. doi:10.1038/s41413-021-00177-7. PMID:35078974
2Martin L*, Cogné B*, Latypova X*, Senaratne LDS*, Koboldt DC, Kellaris G, Fievet L, Le Meur G, Caldari D, Debray D, Nizon M, Frengen E, Bowne SJ; 99 Lives Consortium; Cadena EL, Daiger SP, Bujakowska KM, Pierce EA, Gorin M, Katsanis N, Bézieau S, Petersen-Jones SM, Occelli LM, Lyons LA, Legeai-Mallet L, Sullivan LS, Davis EE, Isidor B.
Mutations in the Kinesin-2 Motor KIF3B Cause an Autosomal-Dominant Ciliopathy. American Journal of Human Genetics. 2020;106(6):893–904. doi:10.1016/j.ajhg.2020.04.005. PMID:32386558
3Tragin M, Degrelle SA, Periou B, Bader-Meunier B, Barnerias C, Bodemer C, Desguerre I, Rodero MP, Authier FJ, Gitiaux C.
Muscle Spatial Transcriptomic Reveals Heterogeneous Profiles in Juvenile Dermatomyositis and Persistence of Abnormal Signature After Remission. Cells. 2025;14(12):939. doi:10.3390/cells14120939. PMID:40558566; PMCID: PMC12190613
4Hou C, Periou B, Gervais M, Martin L, Berthier J, Baba-Amer Y, Souvannanorath S, Lechapt-Zalcman E, Malfatti E, Gherardi RK, Relaix F, Bencze M, Authier FJ.
Interferon-γ causes myogenic cell dysfunction and senescence in immune myopathies. Brain. 2025;148(8):2883–2898. doi:10.1093/brain/awaf153. PMID:40296760
Muscle stem cell-based therapeutic and bioengineering strategies for neuromuscular disorders
Group 3, led by N. Didier, explores the therapeutic potential of muscle stem cells as targets for neuromuscular diseases, as well as their applications in cell therapy and tissue engineering.
One major axis investigates the role of muscle stem cells in spinal muscular atrophy pathophysiology . By combining tissue engineering and multi-omics approaches, as well as animal models, the group focuses in particular on interactions between muscle stem cells, neuromuscular junctions, and motor neurons, with the aim of developing combinatorial therapeutic strategies to maintain neuromuscular function over the long term.
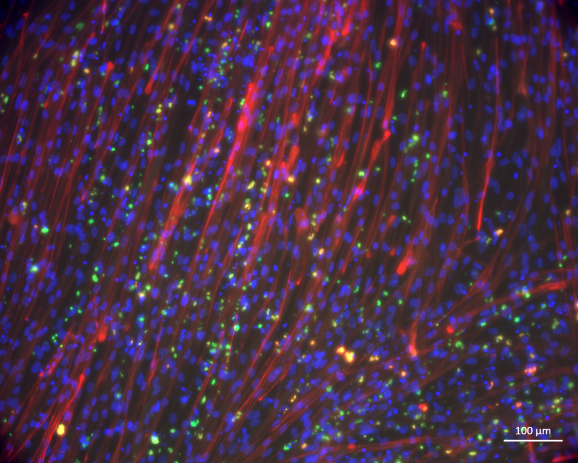
Additional research aims to: 1) Develop pharmacological strategies to preserve the therapeutic potential of human muscle stem cells during in vitro expansion; 2) Engineer 3D muscle models to study the early molecular mechanisms of neuromuscular diseases and test new therapies; 3) Elaborate regenerative medicine strategies for the repair of muscle lesions based on biomimetic scaffolds combined with myogenic cells derived from human muscle stem cells or induced-pluripotent stem cells.
These approaches position muscle stem cells at the core of innovative regenerative and translational therapies for neuromuscular disorders.
Reference:
1Ozturk T, Mignot J, Gattazzo F, Gervais M, Relaix F, Rouard H, Didier N.
Dual inhibition of P38 MAPK and JNK pathways preserves stemness markers and alleviates premature activation of muscle stem cells during isolation. Stem Cell Research & Therapy. 2024;15(1):179.
2Mecca J, Mignot J, Gervais M, Ozturk T, Astord S, Berthier J, Bauché S, Messéant J, Biferi MG, Rouard H, Barkats M, Relaix F, Didier N.
Targeted knockdown of SMN in muscle stem cells induces non-cell autonomous loss of motor neurons. bioRxiv (2024). doi:10.1101/2024.12.19.629483. Accepted in Brain (January 2026).
Tendon and Bone Tissue Engineering
Group 4, led by H. Rouard, focuses on tendon and bone tissue engineering.
Tendons provide the mechanical junction between muscle and bone, but their spontaneous repair is limited by insufficient vascularization. Surgical repair of tendon ruptures is often associated with complications such as restricted movement, scar tissue formation, chronic pain, and secondary ruptures. The group is developing tissue-engineered tendon substitutes to promote robust healing of the osteotendinous junction and to limit postoperative complications.
This work uses adipose tissue–derived stromal cells (ADSCs), which are readily accessible and capable of differentiating into tenocytes. The objective is to generate a biohybrid artificial tendon composed of ADSCs pre-differentiated toward a tendon phenotype and cultured within three-dimensional scaffolds. These constructs will be evaluated in preclinical models of Achilles tendon rupture. Particular attention is given to improving tendon-to-bone anchorage through the use of porous titanium interfaces, developed in collaboration with Group 5 (N. Chevallier).
These preclinical studies are conducted in close collaboration with orthopedic surgeons and benefit from tissue production platforms compliant with GMP standards, with the long-term objective of developing an advanced therapy medicinal product.
Bone Tissue Engineering and Regenerative Medicine
Group 5, led by N. Chevallier.
Our group develops innovative approaches to improve the treatment of large or complex bone defects, which still represent a major clinical challenge. Mesenchymal stromal cells (MSCs) are at the heart of our strategies, and their potential has been demonstrated in several clinical trials (FP7 REBORNE, H2020 Maxibone). Despite these advances, bone healing often remains slow or incomplete, particularly in large or complex geometric reconstructions.
Our research is organized around four main areas:
- Analysis of the mechanisms of bone regeneration induced by MSCs, using animal models and multi-omic approaches. This work aims to identify the cellular and molecular pathways involved in chemotaxis, osteogenic differentiation, angiogenesis, and the establishment of a microenvironment conducive to tissue repair.
- Development of prevascularized bone substitutes derived from a single cellular source. We combine vascularized bone organoids, bioactive biomaterials, and 3D‑printed scaffolds to generate constructs enabling rapid vascular anastomosis, improved post‑implantation cell survival, and homogeneous ossification. These models will also serve as in vitro platforms to study cell–cell interactions in healthy or pathological contexts, as well as responses to therapeutic molecules.
- Establishment of universal protocols based on iPS cells, to generate standardized, safe, and clinically compatible endothelial and osteo‑competent lineages. This includes optimization of differentiation steps, tissue maturation, and quality‑control procedures.
- Facilitating the clinical use of advanced therapy medicinal products (ATMPs) by optimizing production, storage, and logistics processes.
Together, these approaches aim to accelerate bone regeneration, improve the feasibility of complex reconstructions, and make cell and tissue therapies more accessible in clinical settings.
Reference:
¹ Marie T, Cojocaru AI, Coquelin L, Varaillon E, Rouard H, Chevallier N. Directly thawed bone marrow mesenchymal stromal cells retain mechanism of action and support bone clinical translation. Stem Cell Reviews and Reports. 2025; Epub ahead of print. doi:10.1007/s12015-025-11025-0. PMID: 41398484.
² Sanz M, Gjerde C, Gjertsen BT, Ortiz-Vigón A, Sanchez N, Hoornaert A, et al. Bone augmentation of atrophic alveolar ridges using a synthetic bone substitute with mesenchymal stem cells: a randomized, controlled clinical trial. Clinical Oral Implants Research. 2025; Epub ahead of print. doi:10.1111/clr.70025. PMID: 40844304.
 Translational Research in Neuromuscular and Musculoskeletal Diseases
Translational Research in Neuromuscular and Musculoskeletal Diseases