News
News
 November 12, 2025:
November 12, 2025:
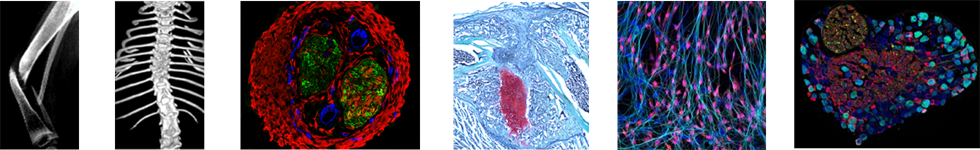
The article “Glial-to-mesenchymal transition of tumor Schwann cells drives the genetic burden in MPNSTs from neurofibromatosis type 1 mouse model” by Radomska KJ, Onfroy A, Lecerf L, Job B, Beaude A, Sesma Sanz L, El Jalkh T, Thieffry D, Charnay P, Wolkenstein P, Ortonne N, Coulpier F, and Topilko P. was published in Science Advances, 2025 Nov 12, Vol 11, Issue 45, DOI: 10.1126/sciadv.adt9210
This work reconstructs the natural history of malignant peripheral nerve sheath tumors, identifies the glial-to-malignant transition as a key driver, and highlights SOX9 as an actionable therapeutic target.
 December 16, 2025:
December 16, 2025:
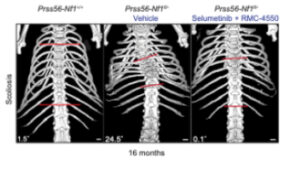
The article “Pharmacological inhibition of RAS pathway alleviates spine deformity in a mouse model of neurofibromatosis type 1” was published in Bone Research, 2025 Dec 16;13(1):103, by Kovaci F, Goachet C, Perrin S, Slimani L, Coulpier F, Tilotta F, Topilko P, Colnot C. DOI: 10.1038/s41413-025-00492-3
In this article, we investigated spinal deformities in the Prss56-Nf1 KO mouse model of neurofibromatosis type 1 (NF1). This is the first NF1 mouse model that recapitulates spinal deformity, along with other NF1 symptoms, providing a clinically relevant model for developing new therapeutic applications. We showed that treating Nf1-KO mice with RAS-MAPK pathway inhibitors prevented spine deformity, highlighting a promising therapeutic strategy for preventing NF1 spine deformity.
 January 29, 2026:
January 29, 2026:
Congratulations to Maria Ethel and Cassandre Goachet for receiving poster prizes at the Bones and Teeth Gordon Conference, Galveston, TX. https://www.grc.org/bones-and-teeth-conference/2026/
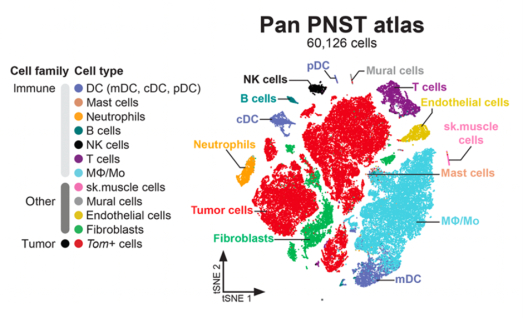
Maria Ethel presented her work on “Human and mouse musculoskeletal stromal cell atlases define skeletal stem/progenitor cell identities and spatial origins”, and Cassandre Goachet on “Distinct epigenetic profiles of skeletal stem/progenitor cells dictate their fate during bone regeneration”.